In 2025, US contract manufacturing deals for FDA-approved drugs posted their steepest decline in five years. Despite record-high US tariffs on EU pharmaceuticals, biopharma companies, including US-based firms, are increasingly choosing to outsource manufacturing for the US market to facilities in Europe rather than shifting work to domestic providers, according to GlobalData, a leading intelligence and productivity platform.
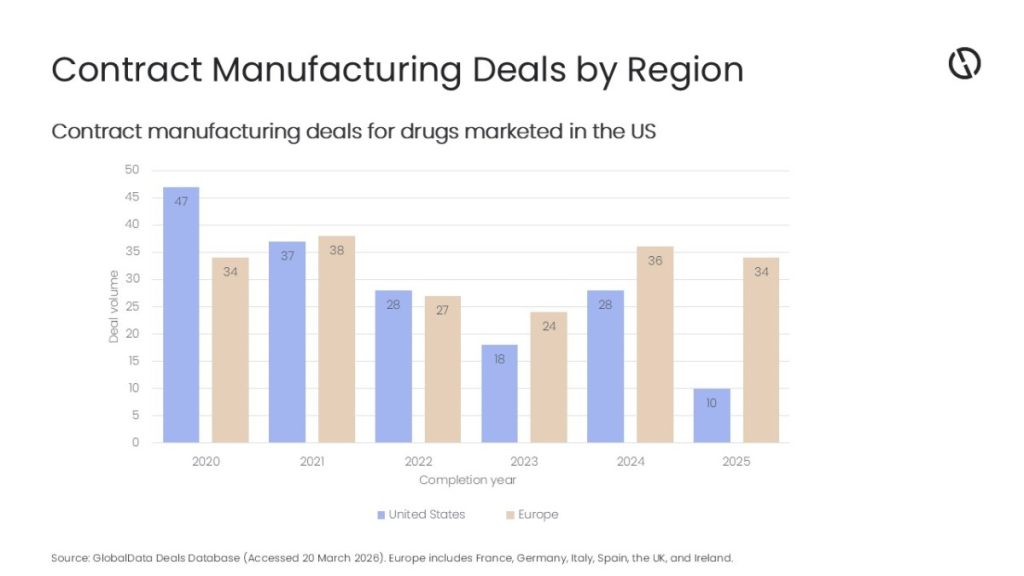
Katia Djebbar, Pharma Analyst at GlobalData, comments: “The gradual decline in US- and Europe-based contract manufacturing deals for FDA-approved drugs between 2020 and 2023 was largely attributed to the rise and subsequent fall in emergency demand for the COVID-19 vaccines over that period. Between 2023 and 2024, deal volume increased in both the US and Europe as the industry recovered post-pandemic.
“In 2025, however, the gap between US- and Europe-based contract manufacturing deals was the widest yet, with Europe recording more than triple the US deal volume. According to GlobalData’s Drugs by Manufacturer database, Germany, which is currently Europe’s leading drug manufacturer, accounted for 12 of those deals, averaging at nine contract manufacturing deals per year for US-market drug manufacturing during 2020-25.”
Undeterred by the 15% US tariffs on EU pharmaceuticals, biopharma companies appear to be increasingly turning to manufacturing in Europe. In 2025, nine out of the 14 US-based pharma companies that outsourced manufacturing, including Johnson & Johnson and Vertex Pharmaceuticals, invested in a total of 13 Europe-based manufacturing deals. By comparison, less than half of these companies invested in US-based facilities, signing a total of eight contract manufacturing deals.
Companies are also following this trend with their in-house manufacturing facilities. Novo Nordisk and Eli Lilly announced investments of $501 million and $3 billion, respectively, to expand their in-house European manufacturing sites. Following the recent FDA approval and success of Wegovy pill, which is currently the first and only oral glucagon-like peptide-1 (GLP1) receptor agonist available in the Western market, Novo is planning to expand its tablets facility in Ireland to meet the current and future US market demand.
Djebbar concludes: “A diversified global supply chain, particularly amid the current unpredictability of the US political climate, will allow biopharma companies to minimize the risk of sudden, catastrophic disruptions to production. The shift in manufacturing deals toward Europe highlights the limited impact that tariffs have had in steering biopharma companies away from European-based contract manufacturing. The region, with Germany in particular, is increasingly becoming an attractive and well-established hub for pharma manufacturing for the US market. Such a shift may hinder the current US administration’s plans to reshore domestic contract manufacturing.”

